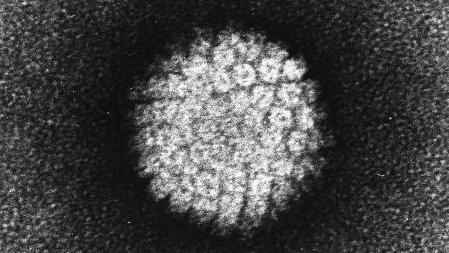
What’s the Latest Development?
In the relatively new global initiative to vaccinate young girls against the human papillomavirus (H.P.V.), the world’s most common sexually-transmitted disease which can also cause cervical cancer, big questions remain about the vaccine’s effectiveness. While there are more than 100 different types of H.P.V., at least 15 of which cause cancer, the vaccine protects only against the two most important cancer-causing strains, HPV-16 and HPV-18. Cervical cancer currently causes 250,000 deaths annually.
What’s the Big Idea?
While clinical trials of an H.P.V. vaccine predict success in reducing rates of cervical cancer, real data on the vaccine’s effectiveness, and potential downsides, will not be available for decades to come. In the mean time, large questions loom: “How long does the vaccine provide protection without a booster? Does it affect natural immunity against H.P.V., and with what consequences? Can we really be sure that the vaccine protects preadolescent girls when proper clinical trials have been carried out only in women aged 16 to 24?”