What is the Universe’s third most common element?
Hydrogen is #1, Helium is #2. Who’s number 3? Hint: it’s not #3 in the periodic table!
“It is the function of science to discover the existence of a general reign of order in nature and to find the causes governing this order. And this refers in equal measure to the relations of man — social and political — and to the entire universe as a whole.” –Dmitri Mendeleev
In the earliest stages of the Universe, it was too hot to form neutral atoms or even atomic nuclei, as they’d immediately be blasted apart by a collision. By time the Universe had expanded and cooled enough that we could form stable nuclei, things were sparse enough that we wound up with 75% hydrogen, 25% helium and just 0.0000001% lithium, with nothing stable beyond that. For tens of millions of years, that’s all the Universe would know, but once we started forming stars, all of that would change.
Today, the Universe is still overwhelmingly hydrogen and helium, but there’s a new #3 in town, and lithium is nowhere close to it. The moment the first star is born, some 50-to-100 million years after the Big Bang, copious amounts of hydrogen start fusing into helium. The percentages of elements in the Universe start tipping away from light elements and towards heavier ones. But if we’re looking for the third most common element, we need to look to the most massive stars: the ones more than about eight times as massive as our Sun.

They burn through that hydrogen fuel very quickly, taking just a few million years to run out of hydrogen in their cores. Once the core is made entirely of helium, it contracts down and starts fusing three helium nuclei into carbon! It only takes approximately a trillion (10¹²) of these heavy stars existing in the entire Universe (which forms about 10²² stars in the first few hundred million years) for lithium to be defeated.

For a very brief amount of time, carbon takes over for lithium as the third most common element in the Universe, but it doesn’t last. You might think carbon will reign forever, since stars fuse elements in onion-like layers. Helium fuses into carbon, then at higher temperatures (and later times), carbon fuses into oxygen, oxygen fuses into silicon and sulphur, and silicon finally fuses into iron. At the very end of the chain, iron can fuse into nothing else, so the core implodes and the star goes supernova.

These supernovae, the steps leading up to them and even their aftermaths, enrich the Universe with all the outer layers of the star, which returns hydrogen, helium, carbon, oxygen, silicon, and all the heavier elements formed through a few other processes:
- slow neutron capture (the s-process), building elements up sequentially,
- the fusion of helium nuclei with heavier elements (creating neon, magnesium, argon, calcium, and so on), and
- fast neutron capture (the r-process), creating elements all the way up to uranium and even beyond.

But we don’t even have just this single generation of stars: we have many. The star systems that are created today are primarily built out of not only the pristine hydrogen and helium, but the leftovers from previous generations. This is important, because without that, we’d never get rocky planets, only gas giants of hydrogen and helium, exclusively!

Over billions of years, the process of star formation and star death repeats itself, although with progressively more and more enriched ingredients. Now, instead of simply fusing hydrogen into helium, massive stars fuse hydrogen in what’s known as the C-N-O cycle, leveling out the amounts of carbon and oxygen (with somewhat less nitrogen) over time.
Additionally, when stars undergo helium fusion to create carbon, it’s very easy to get an extra helium atom in there to form oxygen (and to even add another helium to the oxygen to form neon), something even our paltry Sun will do during the red giant phase.

But there’s one killer move that stars have that makes carbon a loser in the cosmic equation: when a star is massive enough to initiate carbon fusion — a requirement for generating a type II supernova — the process that turns carbon into oxygen goes almost to full completion, creating significantly more oxygen than carbon by time the star is ready to explode.
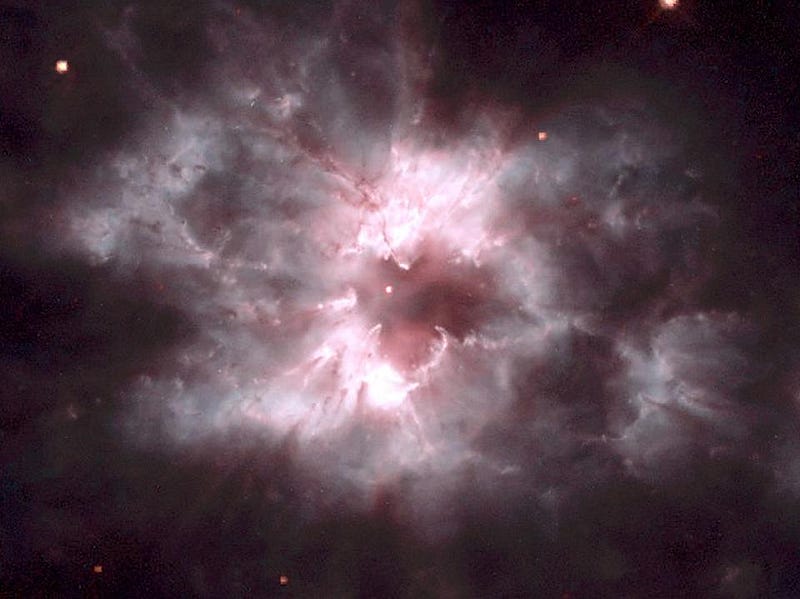
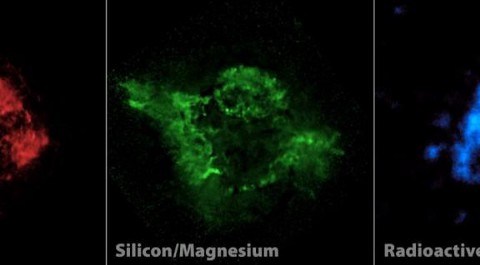
When we look at supernova remnants and planetary nebulae — the remnants of very massive stars and sun-like stars, respectively — we find that oxygen outmasses and outnumbers carbon in each and every case. We also find that none of the other, heavier elements come close!
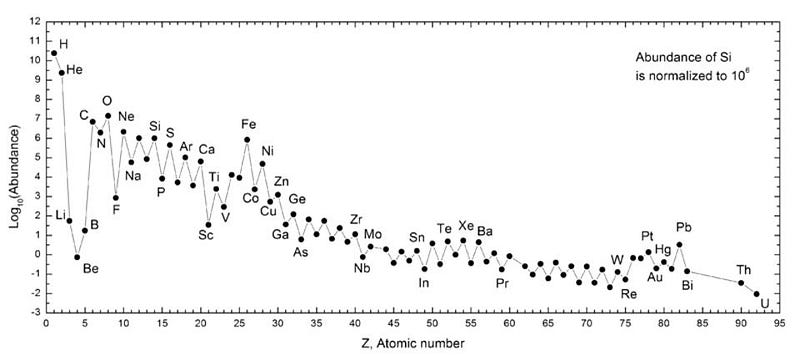
Yes, hydrogen is still #1 by a wide margin, and helium is #2 by a very large amount as well. But of the remaining elements, oxygen is a strong #3, followed by carbon at #4, then neon at #5, nitrogen at #6, magnesium at #7, silicon at #8, iron at #9, and sulphur rounding out the top 10. Lithium? It’s down at about #30 by today.
What will the far future hold? Over long enough time periods, periods that are at least thousands (and probably more like millions) of times the present age of the Universe, stars will continue to form until the fuel is either ejected into intergalactic space, or until its completely burned as far as it can go. When this occurs, helium might finally overtake hydrogen as the most abundant element, or hydrogen may stay #1 if enough of it remains isolated from fusion reactions. Oxygen and carbon will continue to rise in abundance as well, and it’s possible that if things work out just right, one of them will crack the top two.
The most important thing is to stick around, because the Universe is still changing! Oxygen is the third most abundant element in the Universe today, and in the very, very far future, may even have the opportunity to rise further as hydrogen (and then possibly helium) falls from its perch. Every time you breathe in and feel satisfied, thank all the stars that lived before us: they’re the only reason we have oxygen at all!
This post first appeared at Forbes, and is brought to you ad-free by our Patreon supporters. Comment on our forum, & buy our first book: Beyond The Galaxy!