Here on Earth, the water cycle – including its solid, liquid, and gaseous phases – is the top factor in determining what the climate and weather of any particular location is going to be like. Whether frozen at the poles or at high elevations, in the liquid phase in the oceans, or any of Earth’s freshwater stores, or in the gaseous phase as a dissolved component of our atmosphere, where the water both is and isn’t over time determines what forms of life are going to survive and thrive in any region involving our planet’s surface.
But we only have a “water cycle” here on Earth because our temperatures and pressures allow water to exist in all three phases. On hotter worlds, water is always a gas, while on significantly colder worlds, water is always in a solid phase unless it’s crushed down to tremendous pressures. Still, those other worlds have their own forms of rains, snows, and other modes of precipitation. From hot Jupiter worlds where it rains rocks, metal, or even gems to cold, frozen worlds where it snows methane, nitrogen, or even hydrogen, planets revel in a great diversity of types of precipitation.

Here in our own Solar System, we have some clues about the weird properties that planets around other stars might experience. Sure, there’s plenty of water here on Earth and even more on some of the moons of Saturn and Jupiter, but at those large distances from the Sun, it’s only beneath a thick layer of high-pressure ice that liquid water can exist at all. But out on those cold, distant, rocky worlds, all sorts of other frozen chemical compounds, when exposed to direct sunlight, can see them melt, sublimate or boil, becoming liquid or gas, and then precipitate down from the skies later on.
Some worlds are rich in carbon dioxide, which is always naturally a gas here on Earth. However, carbon dioxide becomes dry ice when it freezes, and at intermediate temperatures and the right pressure conditions, can become liquid as well, as has been found in meteorites here within our Solar System. Methane makes solids and liquids on Saturn’s large moon Titan, where it coexists alongside the normal gas phase. And on far-flung Pluto, Triton, and other worlds out there in the Solar System’s far recesses, rains and snows of not only methane but carbon monoxide and nitrogen are thought to be commonplace.

Meanwhile, we have hotter worlds as well. Mercury, being airless, isn’t a great candidate for various types of precipitation, as normally we think about precipitation as occurring where:
- a solid or liquid on the world’s surface either evaporates, sublimates, or boils,
- where it becomes a gas, and rises through the atmosphere depending on its density,
- but as it rises, the temperatures and pressures change,
- and at some point, the gas enters an environment where it will precipitate out of the gaseous phase,
- becoming either liquid droplets or entering the solid phase, where it can remain suspended,
- and when either the droplets or solid structures – including crystalline structures, depending on the molecule – become large and massive enough, they fall as precipitation like rains, snows, or hail.
There’s a hint of this on Venus, where the atmosphere is made largely of carbon dioxide and nitrogen, but the third most common component is sulfur dioxide. That sulfur originates from volcanic activity, and when combined with carbon dioxide and water vapor, can be transformed into sulfuric acid. Venus has thick hazes of sulfuric acid clouds, and when those clouds become massive and heavy enough, they lead to the ultimate in acid rain: sulfuric acid rain, with such a strong acidity that its pH, which is usually measured on a scale ranging from 0 to 14, is about -1.2!
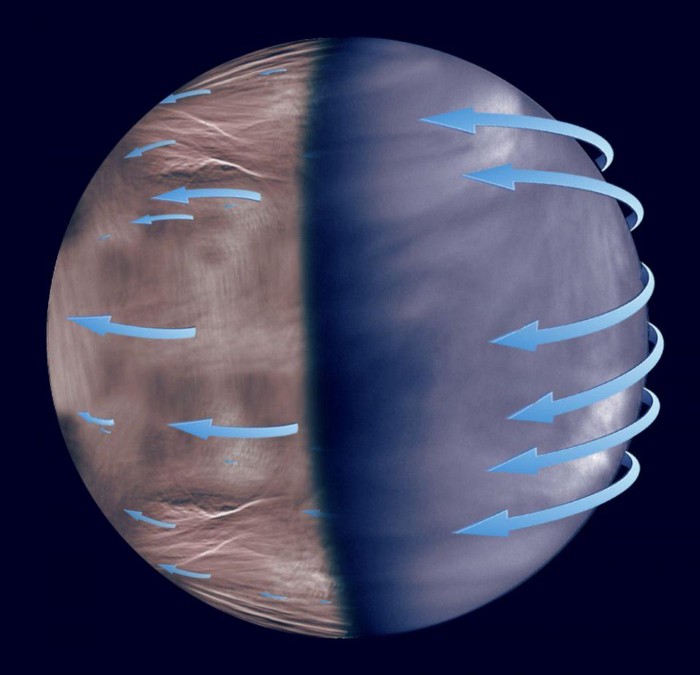
As extreme as Venus is on the hot end in our own Solar System – and as frozen worlds like Triton and Pluto are at the other extremes – there are plenty of places in the Universe with even greater extremes. There are colder worlds than those found in the nearby region of our Kuiper belt, as 95% of all stars are cooler and less luminous than the Sun, while numerous planets have been found at more than 100 times the Earth-Sun distance. At those extremes, rock-and-ice worlds might not only have nitrogen, methane, and carbon monoxide precipitating, but even hydrogen precipitation, while giant worlds might have helium rain as well.
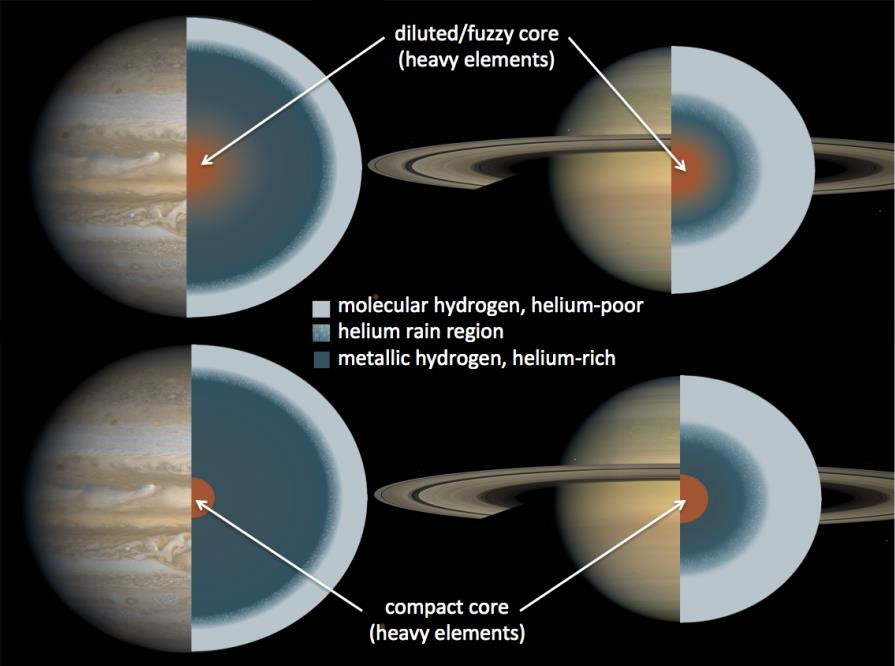
Deep within a giant world like Saturn and Jupiter, and far more common around worlds where the temperatures are lower, the high pressures can transform both hydrogen and helium into the liquid phase. These liquids, due to the vast difference in electronegativities and polarizabilities of these atoms/molecules, will separate much like oil and water do here on Earth, which can cause the helium to precipitate out and sink further. It’s thought that this process can lead to liquid helium rain in the interior of giant planets, and this might be more common the cooler and more massive the planet in question is.
Meanwhile, there are certain to be analogues of ice-and-rock planets at similarly extreme distances from their parent stars, where not only are compounds like nitrogen, methane, and carbon monoxide extremely volatile, but even hydrogen can be as well. Below about 10K, hydrogen is always a solid, but at slightly higher temperatures it can be solid, liquid, or gas dependent on the pressures in its environment. Any world that either has cold nights and warm days, or elliptical orbits that take it nearer-and-farther from its parent star than a critical value will see hydrogen rains and snows in addition to the frozen hydrogen accumulating on its surface.
It’s thought that collisions between such frozen worlds, as well as major impacts upon them, can be responsible for kicking up large fragments of these volatile ices, which can be made primarily of hydrogen, nitrogen, or other abundant but easily-freezable compounds. These icebergs can then wander through the interstellar medium for hundreds of millions or even billions of years, dependent on their sizes, and many of them will eventually come close enough to another star that they can heat up and offgas, accelerating in the process. It’s thought by many that this is precisely where the first interstellar object ever observed from Earth, ‘Oumuamua, originated from.

But there are plenty of worlds that orbit far closer to their parent star than either Mercury or Venus orbit our Sun, and those worlds come in a variety of masses and sizes. While it’s unknown how close a rocky planet can be to its parent star and still have a substantial atmosphere, gas giant worlds can hold onto their gases even with orbital periods of only days or even hours, and can have atmospheric temperatures skyrocket well into the several thousands-of-degrees range. With such extreme temperatures, a wide variety of elements and molecules can be vaporized and launched into the gaseous phase.
Some of those worlds will have a water cycle like Earth does, but that’s where the similarities to Earth end. Instead, their water molecules will be torn apart into individual atoms and then ionized into plasmas on the Sun-facing side: where electrons are stripped off of their atomic nuclei. However, planet-wide winds will circulate those ionized plasmas back on the planet’s night side, where they will cool, recondense into water molecules, and may even freeze and precipitate. The hottest worlds in the entire Universe, in fact, may be extremely rich in nighttime snows.

However, it’s the heavier elements that can lead to the most exotic forms of precipitation of all. On a solid world, elements such as iron and aluminum can be vaporized by the daytime heat and radiation, again becoming heavily ionized in the process. As they drift over to the night side of the planet, they’ll cool and condense, with the possibility that they’ll form clouds in the process as well. Although it sounds strange, depending on the pressure and temperature of the atmosphere, metal clouds can persist under these extreme conditions.
When those clouds become massive enough, they can lead to precipitation as well. While metallic rain is one of many possibilities, another possibility arises as well due to the presence of corundum. Corundum is a chemical compound made of aluminum and oxygen, and it can form with traces of other metals as well: iron, titanium, vanadium, and chromium among them. Corundum is famous on Earth for forming a wide variety of gemstones, and the conditions on hot exoplanets are no different. As exotic as metallic rain sounds, there are likely worlds out there where it rains sapphires and rubies as well.

Rocky planets can also see that surface rock vaporized and launched into the gaseous phase by the intense radiation striking the planet. There are exoplanets that are well known to have lava-rich surfaces, where the external conditions allow surface rock to melt, enter the liquid phase, and flow freely across the surface. These planets are like extreme versions of Jupiter’s innermost Galilean moon, Io, except instead of “only” having to contend with tidal heating from the main body that it orbits, a sufficiently hot exoplanet will experience a tremendous amount of incoming radiation from its parent star.
As lava flows across the exoplanet’s surface, it can boil away, just as any sufficiently heated liquid will enter the gaseous phase (or become super-critical). These rocky materials will rise through the atmosphere and eventually reach the planet’s night side, where they’ll cool and condense once they do. Rocky material will enter the solid phase, where it can actually remain suspended in the atmosphere until the particles reach a certain size. Whether you call them “snow” or “hail” is a matter of personal preference, but there are planets out there in the Universe where sands and rocks precipitate from the skies.

Among the gas giant worlds that orbit extremely close to their parent stars – the “hot Jupiters” of the Universe – a wide variety of clouds and hazes have been seen that would have been wildly unexpected just a few years ago. These include:
- hydrocarbons, including not just methane but much more complex, long-chained molecules,
- salts, including a large abundance of potassium chloride,
- silicates, which represent a lot of the rock-and-sand rain likely to be found among exoplanets,
- as well as sulfurous and metallic features, including aluminum oxide and titanium dioxide.
At the greatest temperature extremes, the elements with the heaviest atomic nuclei can be vaporized and turned into gases and plasmas, where they can participate in their planet’s clouds, hazes, and precipitation cycles. Across all worlds, there’s one key commonality: once a critical mass and density of solids and/or liquids accumulates in a layer of a planet’s atmosphere, precipitation occurs. It’s only a question of the specifics of pressure and temperature gradients and differences, as well as the particular elements and compounds in question, that determine whether what precipitates does so as a solid or liquid.

Even with “only” approximately 5,000 exoplanets discovered as of 2022, there are some incredibly wild and exotic worlds as far as known precipitation goes.
- HAT-P-7b: a Hot Jupiter exoplanet that likely rains sapphires.
- WASP-121b: a Hot Jupiter exoplanet with metallic rain and a plasma-rich water cycle.
- Kelt-9b: the hottest known exoplanet at 4000 °C (7000 °F) with iron-and-lava rain.
- K2-141b: a hot, Earth-sized exoplanet with a rocky surface, liquid rock oceans, and rock-based rain and snow.
- And CoRoT-7b, a possibly rocky larger-than-Earth exoplanet where it rains rocky minerals, including silicates and metals.
Remarkably, all of these worlds have been discovered over the past 30 years, reminding us of just how young and novel the science of exoplanets truly is. With the advent of new telescopes both on the ground and in space, we’re poised to find, identify, and characterize thousands upon thousands of new worlds, and will likely find many types of precipitation that would surprise even the most imaginative among us. When it comes to the Universe, it’s truly amazing what protons, neutrons, and electrons can do under the combined influences of gravity, nuclear and chemical physics, and time.
