From Space, in HD.
“You cannot rob me of free nature’s grace,
You cannot shut the windows of the sky
Through which Aurora shows her brightening face.” –James Thomson
Ever since humanity first broke the chains of gravity and left Earth’s atmosphere, we’ve been able to see our planet for what it actually is: a blue marble floating in the abyss of space. The oceans give our world its primary color, along with the whites from the clouds and ice-covered regions, the lush greens of the tree-covered continents and the browns of the arid, dry desert lands. But when we look at the Earth at night, there’s no reflected sunlight illuminating those common sights, and so the more subtle signals coming from city lights, lightning strikes and atmospheric effects are visible. When the circumstances are just right, most prominent and spectacular of all are the sights of the aurorae.
Aurorae are usually green in color, originating in the upper layers of the Earth’s atmosphere, 50, 100, or even 200 or more kilometers above our planet’s surface. They come down in waves, shimmering across the sky, and form in circles hundreds or even thousands of miles around near the Earth’s poles. From the perspective of someone in orbit around Earth, rather than on the planet’s surface, the aurorae appear as a strange, green fire blazing its way across the border of where the atmosphere ends and space begins.

But there’s a much greater story than fire taking place here. What’s actually taking place is only possible because of the Sun and its interplay with both Earth’s magnetic field and the atoms and molecules in our atmosphere. In fact, of all the worlds in the Solar System, only Earth has the visible-light aurorae in the colors that we do, and it’s because of the unique combination of these three factors.

1.) Solar outbursts: under normal circumstances, the Sun emits a steady stream of particles, known as the solar wind. Superheated gas in the Sun’s corona ionizes the atoms there, and particles — protons, electrons and heavier atomic nuclei — get sent off at around a million miles per hour through the Solar System in all directions. But when a solar flare, coronal mass ejection or other outbursts occur on the Sun, a huge increase in flux occurs in one particular direction, and the particles that fly through the Solar System do so at significantly increased speeds, up to ~0.8% the speed of light! Randomly, a few of those outbursts will be directed at one of the planets in the Solar System, including Earth.

2.) Earth’s magnetic field: the magnetic dynamo in the Earth’s core creates a relatively strong magnetic field that encompasses not only the entire Earth, but that extends all the way out into space. Most of the charged particles that head our way are deflected away from our world by this magnetic field, but the Earth’s two magnetic poles — north and south — are our vulnerable spots. Magnetic field lines can only loop around, and so they have to penetrate through the Earth at two different locations. Where they do, the charged particles, rather than deflecting away from Earth, get funneled down into a circular region surrounding those poles. The stronger the outburst from the Sun, the farther away from the poles the aurorae reach, creating a more spectacular display.

3.) Earth’s atmosphere: and all of this would be moot if we didn’t have the atmosphere that we do. The combination of nitrogen (77%), oxygen (21%) and water vapor (~1%) are enough to create the full gamut of colors that we see. The other components of our atmosphere are either inert (argon, at ~1%) or in such small concentrations (0.04% or less, like for carbon dioxide or methane) that they don’t cause a notable effect. But the charged particles that come in ionize and break apart the atomic and molecular bonds that bind these atmospheric components together, and create these unstable ions and free electrons.
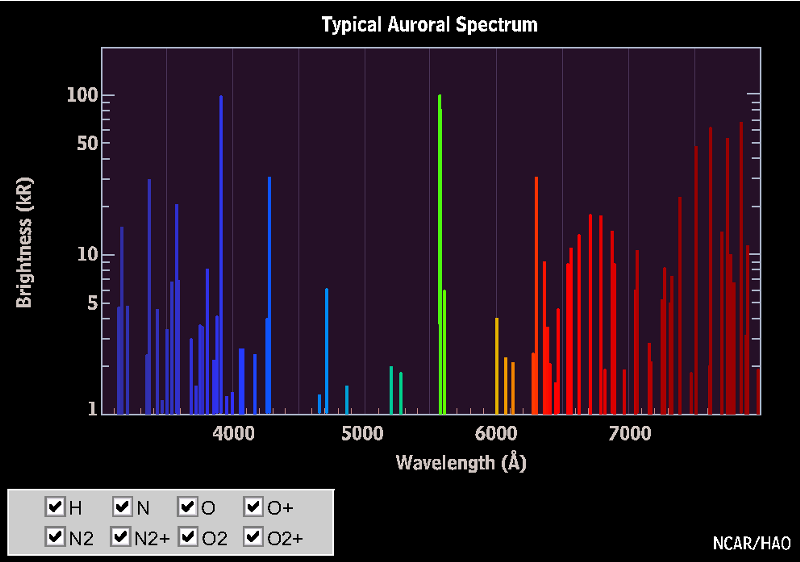
When the free electrons finally find the ions they bind to, they drop down in energy, creating an incredible display of colorful possibilities. Of all of them, it’s the oxygen (mostly, with the strong emission line at 558 nanometers) and the nitrogen (secondary, with the smaller line at a slightly higher wavelength) that create the familiar, spectacular green color we most commonly associate with aurorae, but blues and reds — often at higher altitudes — are sometimes possible, too, with contributions from all three of the major atmospheric elements and their combinations.

A careful observer of the Earth from the ISS will notice other greens, yellows and reds, and that phenomenon is always there: the airglow of our atmosphere. Plain old sunlight is enough to create small amounts of ionization at various layers in our atmosphere, and when the oxygen (green), the sodium (yellow) or the hydrogen (red, higher up) recombine with the electrons at night, causing the steady emission of these colors. In addition, blue lightning strikes and the glow of city lights make the view from the ISS truly one-of-a-kind.

But it’s the combination of those three factors — the Sun’s outbursts, the Earth’s magnetic field and our atmospheric composition — that lead to the spectacular auroral shows that delight skywatchers from both above and below so thoroughly. Only on Earth does this incredible combination occur, and there’s no other world we know of that has shows quite like our own. For the first time, in the highest-resolutions ever, we can now all enjoy the show for ourselves.

This post first appeared at Forbes. Leave your comments on our forum, check out our first book: Beyond The Galaxy, and support our Patreon campaign!
