Too much of the wrong type can be deadly, but not all mercuric compounds are created equal.
Today, we have a guest post courtesy of Adrianne Stone. Adrianne is a graduate student in Translational Biology & Molecular Medicine studying human genetics and the use of gene therapy for the treatment of bone and joint disorders. Her family thinks she’s a huge nerd, but she is, so it’s okay. She is the editor of The Science Collective on Medium, and in her spare time likes to make things with her hands — like jewelry or iPad cases — and blogs about it at yeahimadeit.com. Follow her @AdrianneElayne on Twitter.
“All things are poison and nothing is without poison; only the dose makes a thing not a poison.” –Theophrastus Phillipus Auroleus Bombastus von Hohenheim
Mercury has long captured the attention of our species with its near-mystical quicksilver allure. Ancient alchemists believed it to be an essential element in the transfiguration of base metals into gold, and it was the component that imbued “metal-ness” upon all metals according to Aristotle. India and China used mercury in traditional medicinal remedies for anything from aphrodisiacs to contraceptives, from sedatives to life-extending elixirs. Want to walk on water? Rub some cinnabar (the ore from which most Mercury is obtained) on the soles of your feet. Are you a super old man but want to have more kids? Stir it in your raspberry juice and bottoms up! Mercury also found its way into art and fashion as a component in pigments and felt-making (through a felt-maker’s urine interestingly enough), and vermillion, made from powdered cinnabar, was always in high demand for rituals in South America, often found adorning the ceremonial burial objects of nobles and kings.
Despite its popularity, our ancestors were also aware of its darker side. For example, the Romans used prisoners and other disposable humans to mine the ore, due to the propensity of miners to die from exposure. (This, quite beneficially, spared the state the need of conducting a formal execution.) Lewis Carroll immortalized the neurotoxic effect of mercury in the popular consciousness in his tale Alice’s Adventures in Wonderland through the character of the Mad Hatter, which was inspired by the malady that afflicted men’s hat-makers of the 19th century. Hatters often worked in poorly ventilated rooms, and the hot mercuric nitrate used to manufacture the felt for the hats permeated the air with toxic mercury vapor. The hatters’ shakes, as it was known, caused men to become pathologically shy and irritable, with prolonged exposure leading to hallucinations, eccentric behavior, and a physical tremor that began in the hands and progressed to the rest of the body (often impairing the ability to walk).

Mercury’s notoriety increased in the 1960s and 70s when a series of high-profile poisoning cases occurred, including an episode involving the dumping of waste water from a chemical factory into Minamata Bay, Japan and a catastrophic incident in Iraq. While the Minamata Bay debacle was due to negligence, the Iraqi poisoning was accidental. Drought had severely reduced harvests over from 1969-1970, so it was decided that seed-grain would be imported from the US and Mexico. The seed-grain was coated in a mercury-based fungicide and was intended for planting; however, the shipments arrived after the planting season was over. Instead, an unwitting populace who did not understand the bilingual warnings on the bags ground up and consumed the grain. Surprisingly, warnings in English and Spanish were less than useful to the Arabic and Kurdish speaking peoples that make up the majority of the population in Iraq.
In both poisoning cases, the culprit of these crimes was methylmercury, a compound of mercury, carbon, and hydrogen that has the nasty habit of building up in body tissues and concentrating as you move up the food chain, a phenomenon known as biomagnification. The people affected experienced a spectrum of neurological symptoms including vision, hearing, and speech impairments, issues with coordination, seizures, and in the most severe cases, death. Even more worrisome was the effect mercury exposure seemed to have on the unborn children of women who had been exposed. Even though the women themselves were unharmed by mercury exposure, and these children never consumed the contaminated foods, they displayed permanent profound neurological symptoms including reduced intelligence, convulsions, inability to track movement with the eyes, growth disruptions, and many other ailments symptomatic of cerebral palsy.
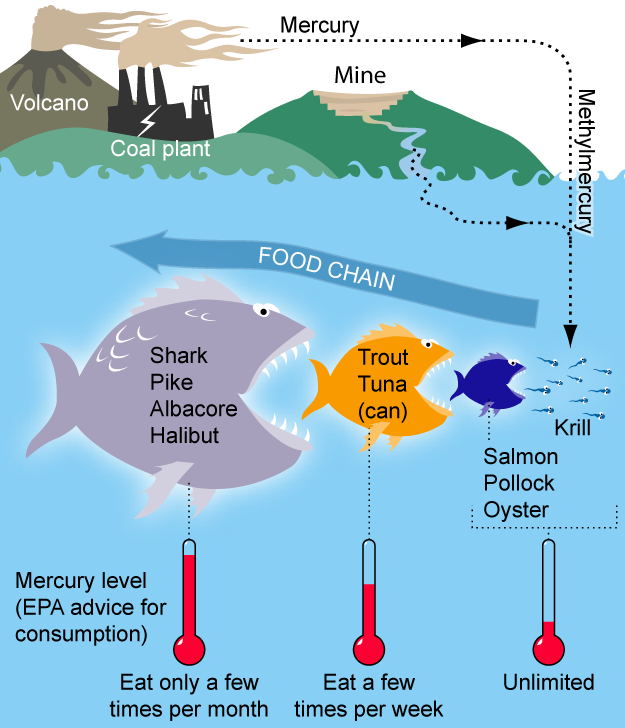
The public was understandably alarmed at the possibility of lurking mercury in the food supply and environment, but most assumed that by limiting their exposure to mercury in their daily lives (i.e. don’t play with it, eat insane amounts of tuna, or work in a mercury mine), the chances of being poisoned were next to nothing. It wasn’t until the publication of a now infamous study correlating MMR vaccinations with an increase in autism in the UK that any connection was made between routine childhood vaccinations, mercury, and autism. In the United States, the furor was kicked off by Congressman Dan Burton, who called for a series of congressional hearings on vaccinations and autism; however, the support groups for parents of children with autism are credited for making the first “connections” between mercury compounds in vaccine preservatives (Thimerosal) and rising autism rates. Incidentally, in the UK, the subsequent uproar was over the MMR vaccine and not the use of Thimerosal, as that mercurial compound was never used in the production of the MMR vaccine, a fact conveniently overlooked in the United States.

Why even put a mercuric compound into vaccines in the first place? Good question. Thimerosal is approximately 50% ethylmercury by weight (note: this is not the same compound as the methylmercury which caused the aforementioned poisonings), and was originally introduced as a vaccine preservative in the early 20th century. Thimerosal, in particular, was the compound chosen for this use because it was found to be both a potent antimicrobial that did not impact the efficacy of the vaccine and also was well-tolerated in humans. Ethylmercury, like methylmercury, is also an organic compound composed of mercury, carbon, and hydrogen; yet the difference in the chemical structure renders it far less toxic and more rapidly excreted from the body.
Prior to the introduction of preservatives like Thimerosal, vaccines had a nasty habit of occasionally becoming contaminated with bacteria or fungi either through the production process, or during administration from multi-dose bottles. In one particularly devastating case in Australia (1928), 12 of 21 children injected from one vial of diphtheria vaccine died from a subsequent staphylococci infection. Thimerosal was shown to be safe for use in humans in several clinical trials before it was used in vaccines, and was for all intents and purposes better than any other preservative available. The scores of people, including children, who were getting severely ill or dying from contaminated vaccines heavily outweighed the low risk associated with the use of the compound in the eyes of healthcare practitioners. When you compared the tens of thousands of lives that were lost annually to vaccine-preventable diseases to the quantitatively much smaller risks of the vaccines themselves, it was a hard position to argue.
Yet who would have expected that 60 years later, this reasoned decision would be derided by vocal anti-vaccination groups as a cold-hearted, moneymaking scheme by a pharmaceutical industry who didn’t care about poisoning children with their toxic vaccines? As many people now know, the vaccine-autism study has been debunked by many follow up studies conducted in the years to follow, and mainstream science does not see any evidence to support the connection Despite this, fears over mercury poisoning from vaccinations have persisted. One reason for this is the confusion over the biological role of ethylmercury (the kind found in thimerosal) as compared to methylmercury (the kind found in the poisoning cases) and the generic use of the word “mercury” to describe them both. While elemental mercury vapor and methylmercury are known to be potent neurotoxins, there is little to no scientific evidence that thimerosal is dangerous at the concentrations found in vaccines (or even scores of times as great).
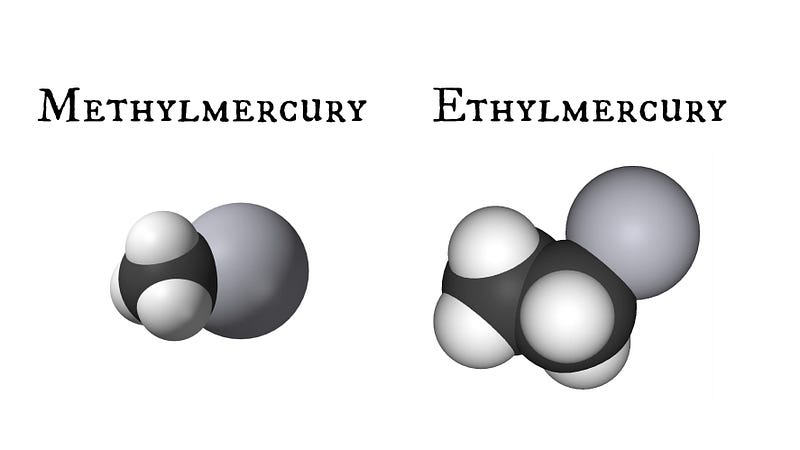
The difference between ethyl- and methylmercury might not seem like much on the surface, but it is very similar to the difference between two common alcohols — the delightful ethanol and the toxic methanol — a critical distinction as any moonshiner will tell you. Methylmercury is much less water soluble and more fat soluble than ethylmercury — making it harder to get rid of, not to mention being a far more potent neurotoxin (200 micrograms/L vs 1000-2000 micrograms/L threshold dose). Studies have also shown that ethylmercury tends to be more quickly processed by the kidneys and excreted than methylmercury, and it takes incredibly high doses of ethylmercury to produce the same kind of neurological defects. That isn’t to say that ethylmercury is completely safe, but then again, neither is ethanol — too much of either compound is going to leave you feeling severely ill.
Unfortunately, due to the lack of exposure guidelines for ethylmercury, the guidelines for methylmercury were used to justify the removal of thimerosal from almost all modern vaccines in the United States. Those guidelines, which were only developed after the mass methylmercury poisonings of the 1960s and 70s — a situation extremely unlikely to ever been seen with ethylmercury — are thought by the overwhelming majority of researchers to be inaccurate. While a good measure of precaution is always appreciated, especially when concerning the safety of medications used in children, using the same standard has only served to increase the confusion and anxiety over the use of Thimerosal, and ultimately led to its usage restriction. Many researchers would have preferred a much less hurried, and more data-driven evaluation of the safety and efficacy of the compound, yet political expediency made this nearly impossible. While the compound has been removed from almost all vaccines in the US, it is still readily used in developing nations, and researchers estimate that replacing it with another preservative will increases costs by two to five times, while also making it harder to transport and refrigerate them. The WHO has stated that requiring its removal would result in many routine childhood vaccines in those nations becoming unavailable, and more children will die from preventable disease as a result.

Fortunately for many of us, there are now many other options for keeping vaccines free of unwanted pathogenic passengers; however, the damage to the public perception of vaccine safety was done. A quick Google search of “mercury poisoning and autism” will turn up thousands of sites that still claim a link between the two, not to mention all manner of conspiracy theories. Though Thimerosal is only now found in some influenza and DTaP vaccines today, the fears of “toxins” and bugaboos in vaccines leading to all manner of illnesses lives on. The controversy over vaccination is one of the best examples of the necessity for people to educate themselves on scientific concepts; there are so many groups with well-intentioned but misguided agendas spreading confusion and misinformation, that it’s difficult for a non-specialist to tease out whose information they should listen to.
A proper understanding of the differences in how the human body responds to and processes different compounds and elements allows people to make informed decisions. No one chooses paint thinner over Johnnie Walker, and no one (in their right mind) would choose to drink methanol-based moonshine. Yet, when it comes to the decision of whether they should vaccinate their children or not, a fear of “mercury” that’s only tenuously connected to scientific facts permeates our world and endangers the lives and health of millions. While the name “mercury” has developed a nasty reputation, its less-toxic ethylmercury form has saved untold numbers of lives, and can continue to do so, so long as we don’t let our ill-founded fears stop us.
Thanks again to Adrianne for a wonderful guest post. If you have comments, feel free to weigh in here or at the Starts With A Bang forum on Scienceblogs!