Proof of ‘God playing dice with the Universe’ found in the Sun’s interior

If not for the indeterminate nature of matter, the Sun would never be able to shine.
“The bedrock nature of space and time and the unification of cosmos and quantum are surely among science’s great ‘open frontiers.’ These are parts of the intellectual map where we’re still groping for the truth — where, in the fashion of ancient cartographers, we must still inscribe ‘here be dragons.’”
–Martin Rees
Deep inside the Sun’s interior, the fusion of lighter nuclei into heavier ones causes a tiny amount of mass to be lost, converted into energy via the famous E = mc². At temperatures of 4,000,000 K or above, all the way up to 15,000,000 K in the Sun’s very center, hydrogen and helium isotopes build their way up to more stable elements, releasing energy and providing all the power that washes over every planet in the Solar System. Yet despite these incredible energies, the protons in the Sun’s core would never be able to begin this chain reaction if the Universe were completely deterministic. It requires the wave nature of quantum mechanics to make it possible, proving that Einstein’s famous statement, that “God does not play dice with the Universe,” was false.

In the 1920s, the world of physics was swept by two major revolutions: General Relativity, which put forth spacetime and the fact that matter and energy curved it as the cause of gravitation, and quantum mechanics, which detailed that all the particles in the Universe also acted like waves. Because of some fundamental properties in quantum physics, it was inherently a non-deterministic theory, meaning that you could only talk about probabilities of certain outcomes occurring, rather than knowing what would result from a particular setup. Two of the most important physicists of the time, Albert Einstein and Niels Bohr, had a number of famous (and public) debates over whether the Universe was inherently deterministic or not, with Einstein arguing yes and Bohr arguing no.

All the way up until his death in the 1950s, Einstein refused to believe, as he called it, that “God played dice” with the Universe. There must be some underlying laws, he reasoned, that determined which particles would behave in which particular fashion, and that it was only a failure of our experimental or observational capabilities that prevented us from seeing the real truth of the matter. When quantum physics was first developed in the 1920s, however, there were only two fundamental forces known: gravity and electromagnetism. The nuclear forces were still unknown, which almost meant that the source of the Sun’s power — nuclear fusion — was unknown as well. If only Einstein knew about this, he could have realized how mistaken he truly was!
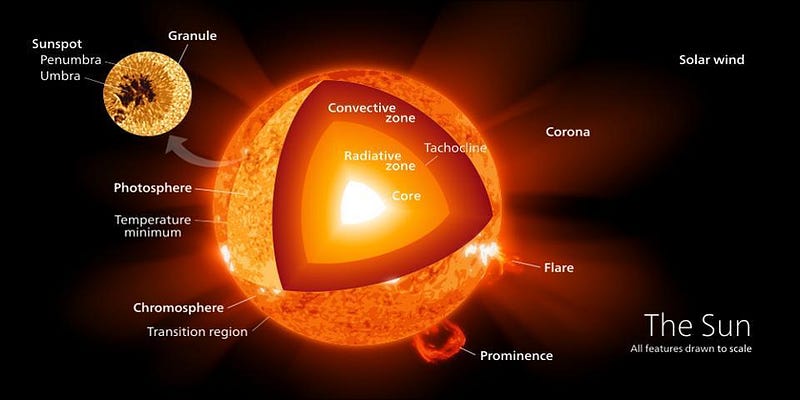
All told, by looking at the power output of the Sun, we measure that it emits a continuous 4 × 10²⁶ Watts, which means that inside the Sun’s core, a whopping 4 × 10³⁸ protons fuse into helium-4 every second. If you consider that there are some 10⁵⁷ particles in the entire Sun, of which a little less than 10% are in the core, this might not sound so far-fetched. After all:
- These particles are moving around with tremendous energies: each proton has a speed of around 500 km/s in the center of the Sun’s core.
- The density is tremendous, and so particle collisions happen extremely frequently: each proton collides with another proton billions of times each second.
- And so it would only take a tiny fraction of these proton-proton interactions resulting in fusion into deuterium — about 1-in-10²⁸ — to produce the necessary energy of the Sun.
So even though most particles in the Sun don’t have enough energy to get us there, it would only take a tiny percentage fusing together to power the Sun as we see it. So we do our calculations, we calculate how the protons in the Sun’s core have their energy distributed, and we come up with a number for these proton-proton collisions with sufficient energy to undergo nuclear fusion.
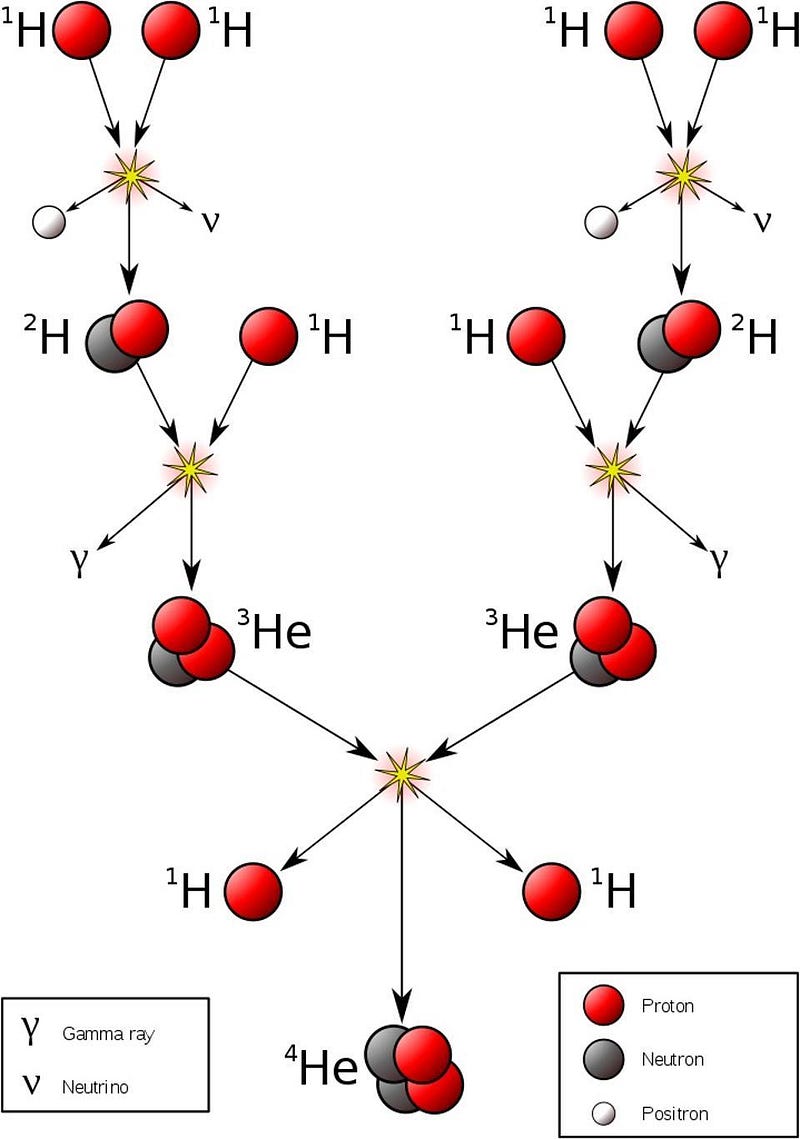
That number is exactly zero. The electric repulsion between the two positively charged particles is too great for even a single pair of protons to overcome it and fuse together with the energies in the Sun’s core. This problem only gets worse, mind you, when you consider that the Sun itself is more massive (and hotter in its core) than 95% of the stars in the Universe! In fact, three out of every four stars are M-class red dwarf stars, which achieve less than half of the Sun’s maximum core temperature.

Only 5% of the stars produced get as hot or hotter as our Sun does in its interior. And yet, nuclear fusion happens, the Sun and all the stars emit these tremendous amounts of power, and somehow, hydrogen gets converted into helium. The secret is that, at a fundamental level, these atomic nuclei don’t behave as particles alone, but rather as waves, too. Each proton is a quantum particle, containing a probability function that describes its location, enabling the two wavefunctions of interacting particles to overlap ever so slightly, even when the repulsive electric force would otherwise keep them entirely apart.
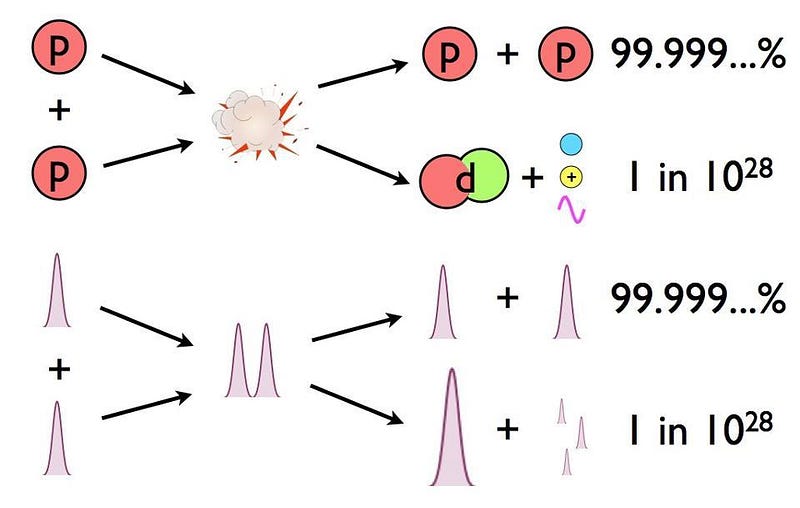
There’s always a chance that these particles can undergo quantum tunneling, and wind up in a more stable bound state (e.g., deuterium) that causes the release of this fusion energy, and allows the chain reaction to proceed. Even though the probability of quantum tunneling is very small for any particular proton-proton interaction, somewhere on the order of 1-in-10²⁸, or the same as your odds of winning the Powerball lottery three times in a row, that ultra-rare interaction is enough to explain the entirety of where the Sun’s energy (and almost every star’s energy) comes from.

If it weren’t for the quantum nature of every particle in the Universe, and the fact that their positions are described by wavefunctions with an inherent quantum uncertainty to their position, this overlap that enables nuclear fusion to occur would never have happened. The overwhelming majority of today’s stars in the Universe would never have ignited, including our own. Rather than a world and a sky alight with the nuclear fires burning across the cosmos, our Universe would be desolate and frozen, with the vast majority of stars and solar systems unlit by anything other than a cold, rare, distant starlight.
It’s the power of quantum mechanics that allows the Sun to shine. In a fundamental way, if God didn’t play dice with the Universe, the nuclear flame that powers the stars would never light, and the life-giving fusion that occurs in our Sun’s core would never come to be. Yet with this randomness, we win the cosmic lottery all the time, to the continuous tune of hundreds of Yottawatts of power. Thanks to the fundamental quantum uncertainty inherent in the Universe, we’ve achieved a chance at existence. Fiat lux.
Ethan Siegel is the author of Beyond the Galaxy and Treknology. You can pre-order his third book, currently in development: the Encyclopaedia Cosmologica.